Trajectory Engineering: Why Medicine Sees Disease Too Late
Most diseases develop across years before they are diagnosed. The future of medicine will depend on learning to see — and influence — those trajectories earlier.
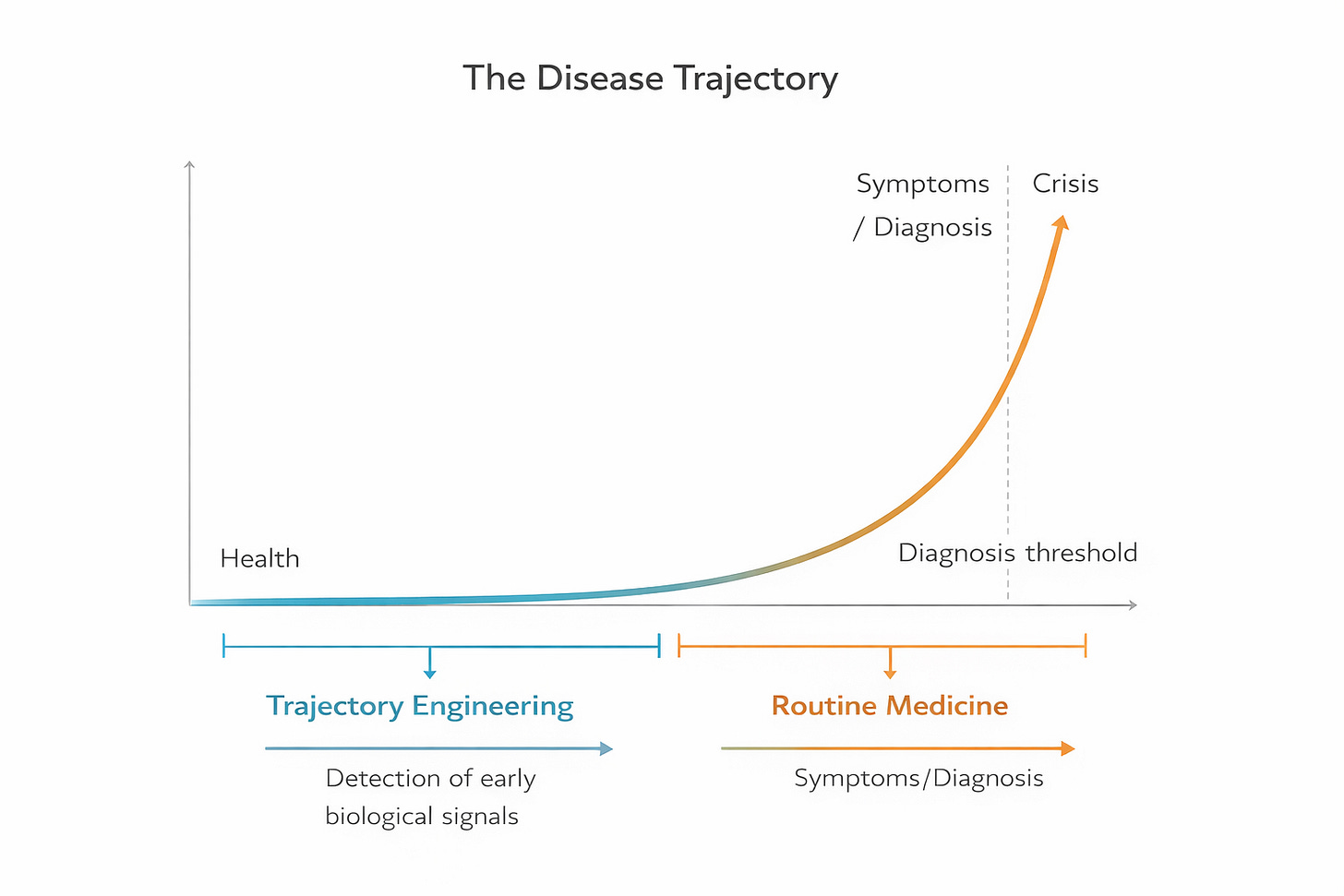
Most diseases do not begin when we diagnose them.
They begin years—sometimes decades—earlier, quietly reshaping biology long before symptoms appear.
Yet modern healthcare systems are organized around the moment when illness finally declares itself.
We wait for the heart attack.
We wait for the tumor.
We wait for the organ failure.
Only then does the full machinery of medicine engage.
That timing problem may be the most important structural challenge in modern healthcare.
Modern medicine is extraordinarily good at responding to crisis.
A patient arrives in the emergency department with crushing chest pain, and within minutes clinicians mobilize imaging, medications, and interventional cardiology teams capable of reopening a blocked coronary artery. Trauma surgeons reconstruct shattered anatomy after catastrophic injury. Oncologists deploy increasingly sophisticated therapies against aggressive tumors. Intensive care physicians sustain life through organ failure that would have been uniformly fatal only a generation ago.
In moments of acute illness, medicine can be remarkable.
Yet there is a quieter truth beneath these achievements: by the time many diseases become visible enough to diagnose, the biological processes behind them have often been unfolding for years—sometimes decades.
The moment of diagnosis feels sudden.
The disease rarely is.
Most of what modern medicine confronts as illness is the late expression of a much longer biological story.
Understanding that simple fact changes how we think about the future of healthcare.
Most disease develops silently for years before symptoms appear. Modern healthcare systems typically intervene after the diagnosis threshold. Trajectory engineering focuses on detecting and influencing these earlier phases
The Illusion of the Medical Event
Healthcare systems, particularly in the industrialized world, are organized around events.
A diagnosis.
A hospital admission.
A surgical procedure.
A course of chemotherapy.
These moments are visible. They are measurable. They are documentable. They fit neatly into billing systems, quality metrics, and clinical records.
Entire infrastructures have been constructed around these events. Hospitals are organized around procedures and admissions. Insurance systems reimburse episodes of care. Quality reporting focuses on outcomes tied to interventions.
But biology does not operate in events.
It operates in trajectories.
Atherosclerosis—the process that eventually produces heart attacks and strokes—often begins quietly in adolescence. Insulin resistance can develop ten or fifteen years before a patient is diagnosed with diabetes. Cancer evolves through the gradual accumulation of genetic mutations and microenvironmental changes long before a tumor becomes visible on imaging. Neurodegenerative diseases such as Alzheimer’s may begin decades before memory symptoms emerge.
The moment we recognize as disease is often the final chapter of a much longer process.
This mismatch between the biology of disease and the structure of healthcare systems shapes much of what we experience as modern medicine.
Medicine Meets Disease Late
The structural reality of healthcare is that it frequently encounters disease late in its trajectory.
Physicians are asked to make consequential decisions at the moment when options are already narrowing. A patient presents with advanced illness and must quickly weigh complex interventions, risks, and tradeoffs under conditions of stress, uncertainty, and fear.
Consent may exist.
But the range of meaningful choices has often already compressed.
This dynamic is not the result of negligence or indifference. It reflects the limits of visibility that historically defined medicine. For most of human history, clinicians could only see disease once it declared itself through symptoms.
The physician’s tools were the patient’s story, the physical exam, and a small set of diagnostic instruments. Disease that had not yet produced symptoms was largely invisible.
Under those conditions, the organization of medicine around episodes of illness made sense.
You treated what you could see.
But that world is changing.
The Rise of Chronic Disease
Another structural shift has intensified the importance of this timing problem.
Infectious diseases once dominated global mortality. Many of those illnesses progressed rapidly and produced clear clinical events. Antibiotics, vaccines, sanitation, and public health interventions dramatically reduced the burden of infectious disease throughout the twentieth century.
Today, however, chronic diseases dominate healthcare.
According to the U.S. Centers for Disease Control and Prevention, chronic diseases account for approximately 90 percent of total healthcare spending in the United States. Cardiovascular disease, cancer, diabetes, chronic lung disease, and neurodegenerative disorders represent the largest sources of morbidity and mortality across industrialized societies.
Unlike many infectious diseases, chronic diseases unfold slowly.
They develop across years or decades through interactions among genetics, environment, behavior, metabolism, and aging. The clinical events we recognize—heart attacks, strokes, cancers, organ failure—are often the final stage of long biological trajectories.
Modern healthcare systems therefore spend enormous resources responding to the late consequences of processes that began long before the system encountered them.
A New Kind of Visibility
For most of medical history, the late appearance of disease was unavoidable.
The biological processes unfolding inside the human body were simply inaccessible until they produced symptoms. Physicians could not see molecular changes, subtle physiological shifts, or early structural alterations.
That limitation is now beginning to disappear.
New forms of data are illuminating earlier stages of disease development in ways that would have been unimaginable even a generation ago.
Genomic sequencing can reveal inherited risks long before illness appears. Continuous monitoring through wearable devices can track heart rhythms, sleep patterns, metabolic signals, and physical activity across months and years. Advanced imaging techniques can detect structural changes in tissues long before symptoms emerge. Longitudinal electronic health records capture patterns that only become visible when data is examined across time.
Artificial intelligence and statistical modeling add another layer of capability. Machine learning systems can integrate vast streams of heterogeneous data—genomics, imaging, physiology, clinical records, environmental signals—and detect patterns that would be impossible for any individual clinician to recognize.
For the first time in human history, medicine is beginning to see the early signals of disease trajectories before those trajectories reach the point of crisis.
This emerging visibility represents one of the most profound shifts in the history of healthcare.
But technology alone does not change systems.
Why This Moment Is Different
The reason trajectory engineering can emerge now is simple: until recently, the necessary visibility did not exist.
For most of human history, the mechanisms of disease were largely invisible. We did not understand the molecular biology of cancer, the metabolic pathways behind diabetes, or the genetic and environmental interactions shaping cardiovascular disease. Medicine worked primarily with symptoms because symptoms were the only reliable signals available.
Over the past several decades, that has begun to change. Advances in molecular biology, genomics, imaging, and computational science have revealed the mechanisms through which disease develops across time. At the same moment, new technologies—continuous sensing, digital health records, and artificial intelligence—allow those signals to be observed and interpreted at scale.
For the first time, scientific understanding of human biology is converging with technological capacity to monitor and analyze health continuously.
That convergence creates the conditions for a new discipline: trajectory engineering—the deliberate effort to detect and influence the arc of health before disease reaches the stage of crisis.
The Concept of Trajectory Engineering
The deeper opportunity lies in what we do with this new visibility.
This is where a new discipline begins to emerge: trajectory engineering.
Trajectory engineering is the discipline of designing health systems that recognize and influence disease trajectories earlier.
Instead of organizing care primarily around the moment when disease becomes acute, trajectory engineering asks a different question:
How can we detect signals of change before disease declares itself?
This shift involves more than new diagnostic technologies. It requires rethinking how medicine defines success.
In the current model, success is often measured by how effectively clinicians respond to disease once it appears. Surgical outcomes, survival rates, hospital readmissions, and complication rates dominate quality metrics.
In a trajectory-based model, success would increasingly be defined by the ability to alter the arc of disease before it reaches the stage of crisis.
Preventing the heart attack.
Detecting cancer while it is still biologically early.
Stabilizing metabolic dysfunction before diabetes develops.
Slowing neurodegeneration before cognitive decline becomes irreversible.
The aim is not merely to treat disease effectively, but to influence the biological path leading toward disease.
Trajectory engineering moves medicine upstream.
Time as a Medical Resource
One of the most important consequences of earlier visibility is something surprisingly simple: time.
When disease is recognized late, decisions must be made quickly. Patients and clinicians face compressed timelines in which complex choices must be made under pressure. Treatment options may be limited by the stage of disease.
Earlier recognition changes that dynamic.
When signals of risk are detected earlier in the trajectory of disease, patients gain time to understand their situation, consider alternatives, and align medical decisions with their values and lives.
Clinicians gain time to intervene gradually rather than urgently. Preventive measures, behavioral adjustments, pharmacologic therapies, and monitoring strategies can be introduced before the biological trajectory has hardened into crisis.
Time expands the range of possible futures.
In that sense, trajectory engineering is not simply about technology. It is about creating conditions in which human decision-making becomes more thoughtful and less reactive.
The Limits of Rescue Medicine
None of this diminishes the extraordinary achievements of modern rescue medicine.
The ability to intervene successfully in acute illness remains one of the greatest accomplishments of contemporary healthcare. Trauma surgery, organ transplantation, intensive care, and advanced oncology have saved countless lives.
But rescue medicine operates at the downstream end of the disease trajectory.
By the time a patient arrives in an operating room or intensive care unit, the biological processes that produced the crisis are already far advanced.
No matter how skilled the intervention, the opportunity to influence the earlier phases of disease has largely passed.
Trajectory engineering does not replace rescue medicine. It complements it by addressing the earlier phases of disease where the trajectory can still be altered.
In the best possible system, rescue medicine becomes the safety net rather than the primary strategy.
The Shift in Perspective
Stepping back, the argument is actually quite simple.
Most diseases develop slowly across time.
Yet healthcare systems still organize their attention around the moment when illness finally becomes undeniable.
That mismatch—between the biology of disease and the structure of medicine—explains much of the strain we see across modern healthcare systems.
Trajectory engineering begins by closing that gap.
Instead of asking only how to respond to disease once it appears, the system begins asking how to detect and influence the biological trajectory much earlier.
Technology Is Not the Limiting Factor
One of the striking realities of modern healthcare is that many of the necessary technological capabilities already exist.
Genomic analysis can identify individuals at elevated risk for certain cancers and cardiovascular conditions. Continuous glucose monitors allow early detection of metabolic dysregulation. Advanced imaging can reveal vascular changes long before symptoms arise. AI-assisted analysis can identify patterns in health data that predict future illness.
The scientific foundations of trajectory-based medicine are advancing rapidly.
Yet the widespread implementation of these capabilities remains uneven.
The limiting factor is not technology alone.
It is the structure of the system in which that technology operates.
Healthcare systems still organize attention, resources, and measurement primarily around the treatment of established illness. Clinical workflows, reimbursement models, and quality metrics largely reward interventions performed after disease becomes clinically apparent.
Incentives shape behavior.
And behavior shapes systems.
Designing the Next Phase of Medicine
The future of healthcare will therefore depend not only on scientific innovation but on institutional design.
Trajectory engineering requires systems that encourage earlier engagement with disease trajectories. It requires clinical workflows that incorporate longitudinal monitoring rather than episodic encounters. It requires data systems capable of integrating diverse signals across time.
Most importantly, it requires aligning incentives with the goal everyone claims to support: sustained health.
When systems reward maintaining health, attention naturally moves upstream.
When they reward intervention after illness, attention remains downstream.
The technologies enabling trajectory visibility are advancing rapidly. Artificial intelligence, genomic medicine, continuous monitoring, and predictive analytics are expanding the horizons of what medicine can detect and interpret.
But technological capability alone cannot reshape healthcare.
The architecture of the system must evolve alongside it.
A Different Future
Imagine a healthcare system in which the first signal of disease is not a heart attack but a subtle change in vascular biology detected years earlier.
Imagine oncology that routinely identifies cancers while they are still microscopic rather than after tumors become visible on imaging.
Imagine metabolic disease managed through continuous physiological insight rather than diagnosed after years of silent progression.
In such a system, the dramatic moments of rescue medicine would become less frequent—not because medicine has become less capable, but because the trajectory of disease has been altered earlier.
Health would increasingly be maintained rather than restored.
This is the promise of trajectory engineering.
Looking Ahead
The scientific capabilities necessary for trajectory-based medicine are emerging rapidly.
What remains is the harder work of redesigning the structures that govern healthcare—how clinicians practice, how data flows, how decisions are made, and how incentives are aligned.
Those structural questions will determine whether the extraordinary technologies now emerging in medicine transform the future of health or simply add complexity to existing systems.
Health is rarely lost in a single moment.
It is shaped over time by signals we notice early—or miss.
Trajectory engineering is the effort to notice earlier.
Next Week
In Part 2, we will explore a difficult question:
Why do modern healthcare systems still reward the moment of crisis rather than the prevention of decline?
And what would it take to change that architecture?
How would we pay for it?
Health 4.0 | Doctor AI | robinblackstone.com
Doctor AI: Reimagining Health — Rebuilding Trust, Architecting Health 4.0
Launches April 7 — Available across all formats on Amazon.